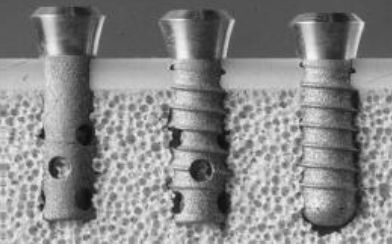
These implants have had an SLA® surface since 1997 and a SynOcta® abutment connection since 1999. They basically correspond to the Straumann® Tissue Level Implant System available today. Since 2005 the implants have been optionally provided with an SLActive® surface, which is scientifically proven to accelerate osseointegration even in compromised patients. Since 2009 all implants have been made of the extra strong titanium-zirconium alloy Roxolid®.

The Straumann® Tissue Level Implant Line
This iconic implant was designed for one-stage surgery to simplify handling and reduce treatment complexity
Basic research for the first Tissue Level generation was done by a group led by the Swiss dentist Prof. Dr. André Schroeder (University of Bern, Switzerland). Along with Per-Ingvar Brånemark, Schroeder was one of the fathers of modern oral osseointegration2. One of Schroeder’s scholars at the time, Prof. Dr. Daniel Buser, was a co-developer and the first clinical user of the ITI Bonefit® system, which led to the modern Tissue Level concept.
Clinical concept
From the very beginning two-part ITI Bonefit® and today’s Straumann® Tissue Level implants were designed for a one-stage surgical protocol. This involves transgingival healing, which eliminates the need for a second-stage surgery to expose the implant shoulder. In comparison with bone level implants, impression-taking and the handling of healing caps, abutments and prosthetic superstructures are significantly more straightforward, making daily clinical practice significantly easier. The protocol also saves time and reduces the complexity of the overall treatment.
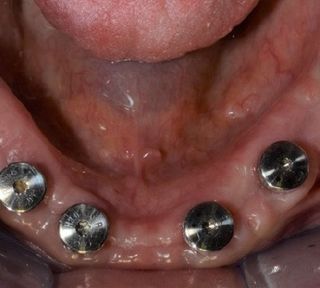
Tissue stability – minimally invasive