With these design features adapted from the Straumann® BLX Implant System, high primary stability can be achieved even in relatively soft bone3. Due to the biomechanical strength of the titanium-zirconium alloy Roxolid®, narrow 3.75 mm diameter Straumann® TLX implants can be used for all indications, including posterior replacements. The combination of maximum primary stability and reduced implant dimensions allows for non-augmentation implantology, even in many cases with minimal bone dimensions4. At the same time, the number of sessions and overall treatment time can be reduced, thereby increasing patient satisfaction and practice efficiency.

Features and benefits
Straumann® developed the TLX Implant System for efficient protocols, both from a surgical and prosthetic point of view.
Clinical versatility and ease of use
Analogous to the original Tissue Level, Straumann® TLX implants can be used in a wide range of clinical indications, including immediate protocols and the rehabilitation of periodontally or systemically compromised patients. In most cases a transgingival protocol can be used, enabling many patients to receive a “one-stage” treatment, i.e., with only one surgical intervention. This applies especially to situations with adequate preoperative bone dimensions and a thick gingival biotype.

The new TLX in the eyes of international experts
What do international experts in implantology think about the new Straumann® TLX Implant System?
Indications for the TLX include single posterior gaps, wider edentulous spaces, free-end situations and edentulous ridges, all of which can be restored in a convenient and predictable way5, 6. The wider diameter of the TLX implant shoulder in comparison with the endosteal part makes it easier to create a natural emergence profile of the crown, even in combination with narrow implants (Figs. 1-3)7. In small gaps a Straumann® TLX narrow neck implant with parallel transgingival collar design can be used.
Learn more about the potentials of the Straumann® TLX Implant System:
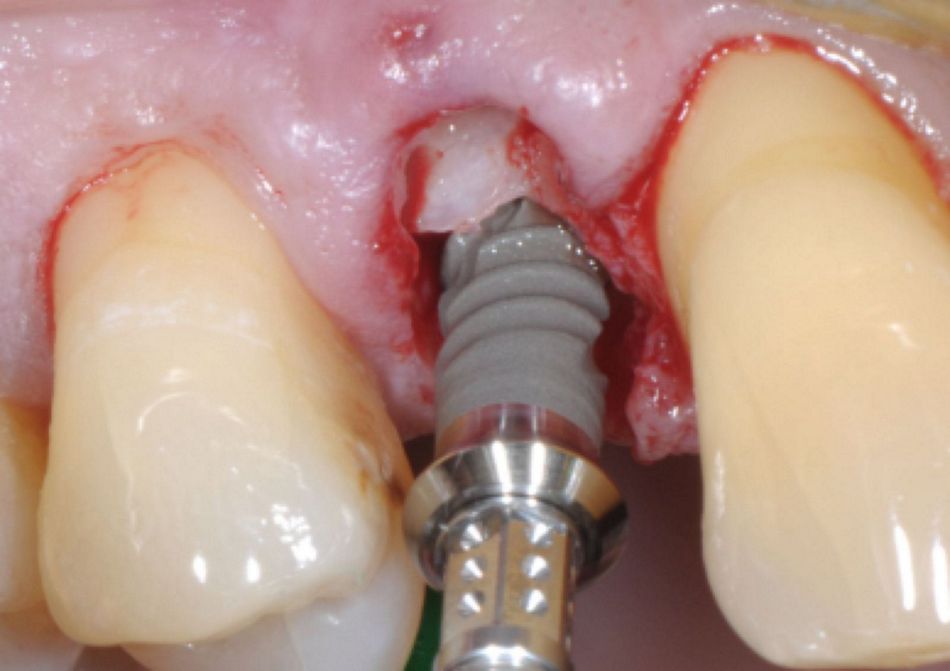
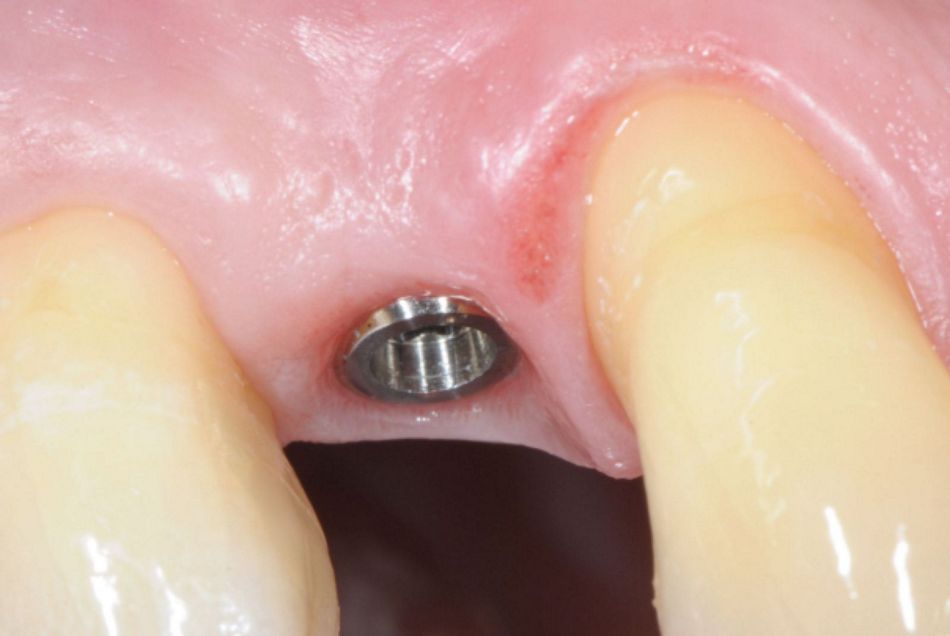
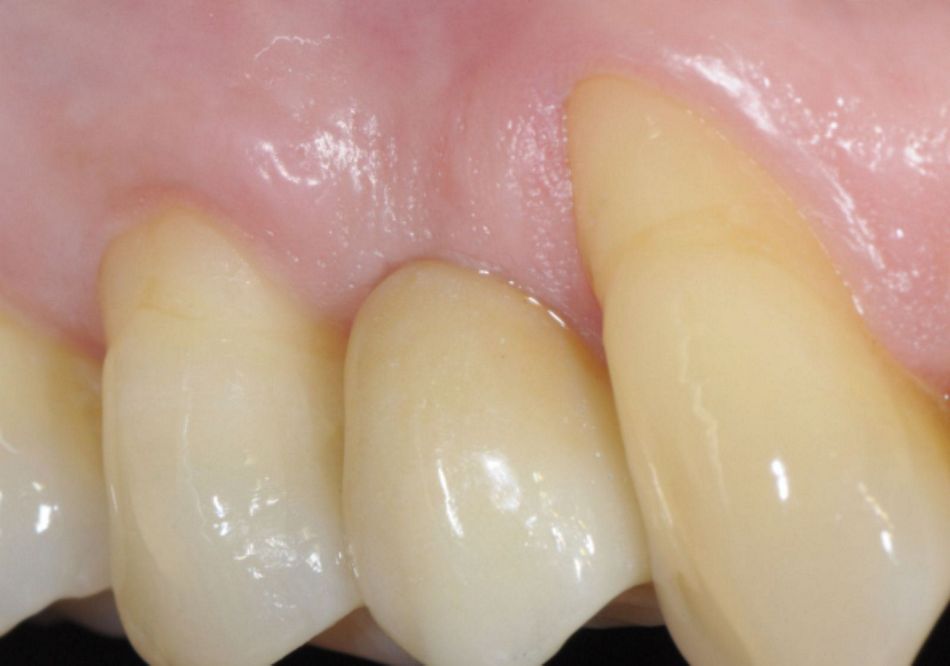
- Figs. 1: A Straumann® TLX implant (Standard platform, 3.75 x 12.00 mm) is placed with the machined-microrough transition at the alveolar crest. After buccal hard and soft tissue augmentation (botiss® biomaterials) and healing, the implant shoulder is positioned in an ideal relation to the gingival margin, producing a highly esthetic result.
- Figs. 2: A Straumann® TLX implant (Standard platform, 3.75 x 12.00 mm) is placed with the machined-microrough transition at the alveolar crest. After buccal hard and soft tissue augmentation (botiss® biomaterials) and healing, the implant shoulder is positioned in an ideal relation to the gingival margin, producing a highly esthetic result.
- Figs. 3: A Straumann® TLX implant (Standard platform, 3.75 x 12.00 mm) is placed with the machined-microrough transition at the alveolar crest. After buccal hard and soft tissue augmentation (botiss® biomaterials) and healing, the implant shoulder is positioned in an ideal relation to the gingival margin, producing a highly esthetic result.
Photos (Figs. 1-3) courtesy of Dr. Mario Rocuzzzo, DT Francesco Cataldi (both Italy) – see case report here
Like the Straumann® BLX, the TLX Implant System is equipped with the TorcFit™ internal conical connection. This interface allows simple handling of prosthetic components, including placement of abutments at the soft tissue level. Direct-to-implant restoration without abutments is also an option (Figs. 4-6), e.g., for the immediate restoration of edentulous ridges with the Straumann® ProArch concept. According to many clinicians’ daily experience the transgingival collar of the TLX effectively promotes soft tissue adaptation and healing around caps or immediate restorations (Figs. 2,5).
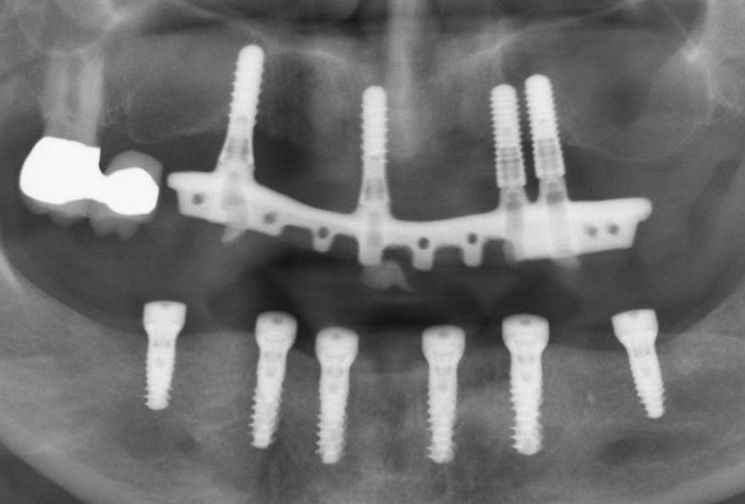
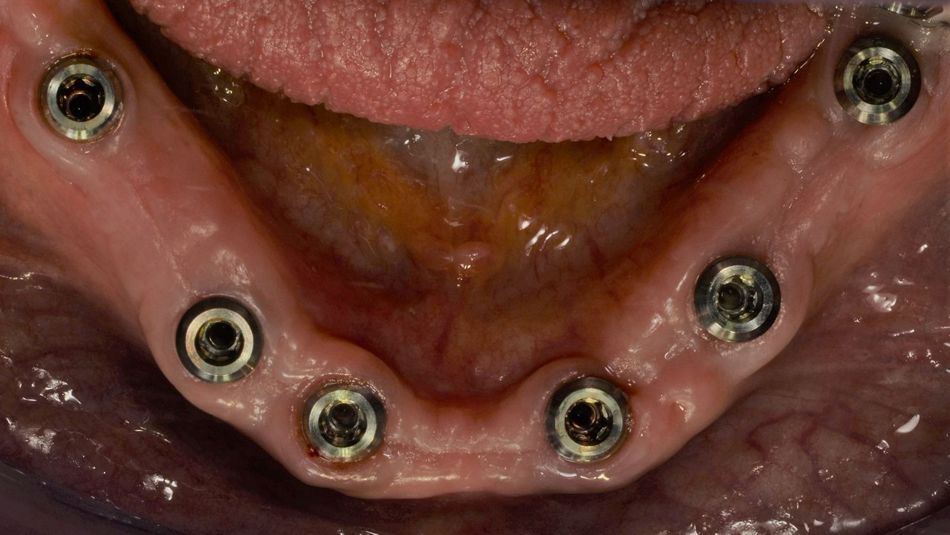
- Fig. 4: Six Straumann® TLX implants have been inserted in the mandible to support a screw-retained CADCAM bridge. The panoramic radiograph detail shows the situation after the fitting of healing abutments and suturing.
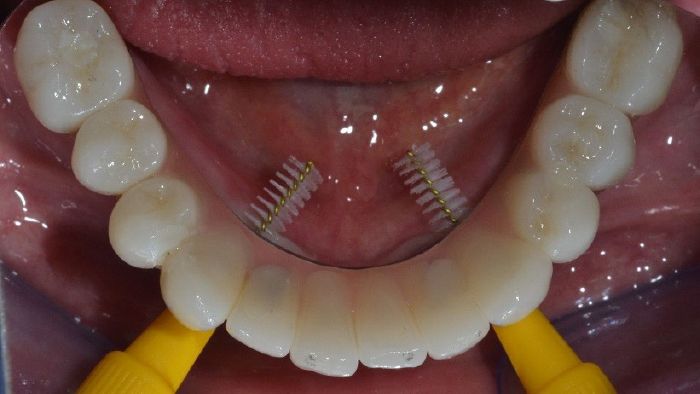
- Figs. 5: After removal of the healing abutments, the soft tissues are free of inflammation and ready for tissue level impression taking. The final restoration was carried out with single ceramic crowns cemented onto a titanium CADCAM framework.
- Figs. 6: After removal of the healing abutments, the soft tissues are free of inflammation and ready for tissue level impression taking. The final restoration was carried out with single ceramic crowns cemented onto a titanium CADCAM framework.
Photos (Figs. 4-6) courtesy of Dr. Eirik Salvesen, Stavanger (Norway) – see case report here
Long-term evidence and ongoing studies
The technology behind the Straumann® TLX Implant System rests on a solid foundation of scientific evidence. It starts with the high-strength implant material Roxolid®, which is composed of 85% titanium and 15% zirconium. In combination with the hydrophilic SLActive® surface, implants manufactured from Roxolid® have shown excellent survival and tissue stability, e.g., in narrow-diameter implants used for mandibular overdentures after 5 years8, 9. These implants have also proven successful in patients with type 2 diabetes mellitus10.
Concerning the Straumann® TLX Implant System itself, a preclinical study was carried out to define the design of ongoing clinical investigations11. A case report was published showing successful immediate loading of a maxillary first molar12. Finally, multicenter clinical studies are underway in more than 10 countries worldwide to further prove the clinical applicability of the system.

How to become an expert
The Straumann® TLX Implant System is built on a strong basis of scientific studies and educational material.
References:
- Kang MH, Jung UW, Cho KS, Lee JS. Retrospective radiographic observational study of 1692 Straumann tissue-level dental implants over 10 years. II. Marginal bone stability. Clin Implant Dent Relat Res 2018;20:875-881.
- Kim S, Jung UW, Cho KS, Lee JS. Retrospective radiographic observational study of 1692 Straumann tissue-level dental implants over 10 years: I. Implant survival and loss pattern. Clin Implant Dent Relat Res 2018;20:860-866.
- Torroella-Saura G, Mareque-Bueno J, Cabratosa-Termes J, Hernandez-Alfaro F, Ferres-Padro E, Calvo-Guirado JL. Effect of implant design in immediate loading. A randomized, controlled, split-mouth, prospective clinical trial. Clin Oral Implants Res 2015;26:240-244.
- Schiegnitz E, Kämmerer PW, Hellwich P, König J, Sagheb K, Al-Nawas B. Treatment concepts of horizontally deficient ridges-A retrospective study comparing narrow-diameter implants in pristine bone with standard-diameter implants in augmented bone. Clin Oral Implants Res 2021;32:1159-1167.
- Fischer K, Stenberg T. Prospective 10-year cohort study based on a randomized, controlled trial (RCT) on implant-supported full-arch maxillary prostheses. part II: prosthetic outcomes and maintenance. Clin Implant Dent Relat Res 2013;15:498-508.
- Derks J, Hakansson J, Wennstrom JL, Tomasi C, Larsson M, Berglundh T. Effectiveness of implant therapy analyzed in a Swedish population: early and late implant loss. J Dent Res 2015;94:44S-51S.
- Sanda M. Four reasons to choose tissue-level implants for posterior sites. ITI blog https://blog.iti.org/clinical-insights/four-reasons-to-choose-tissue-level-implants-for-posterior-sites/. https://blog.iti.org/: International Team for Implantology (ITI), 2022.
- Quirynen M, Al-Nawas B, Meijer HJ, Razavi A, Reichert TE, Schimmel M, et al. Small-diameter titanium Grade IV and titanium-zirconium implants in edentulous mandibles: three-year results from a double-blind, randomized controlled trial. Clin Oral Implants Res 2015;26:831-840.
- Muller F, Al-Nawas B, Storelli S, Quirynen M, Hicklin S, Castro-Laza J, et al. Small-diameter titanium grade IV and titanium-zirconium implants in edentulous mandibles: five-year results from a double-blind, randomized controlled trial. BMC Oral Health 2015;15:123.
- Cabrera-Domínguez J, Castellanos-Cosano L, Torres-Lagares D, Machuca-Portillo G. A Prospective Case-Control Clinical Study of Titanium-Zirconium Alloy Implants with a Hydrophilic Surface in Patients with Type 2 Diabetes Mellitus. Int J Oral Maxillofac Implants 2017;32:1135-1144.
- El Chaar E, Puisys A, Sabbag I, Bellón B, Georgantza A, Kye W, et al. A novel fully tapered, self-cutting tissue-level implant: non-inferiority study in minipigs. Clin Oral Investig 2021;25:6127-6137.
- Morton D, Orgev A, Polido WD. Immediate Loading of Straumann(R) TLX Implant in Grafted Maxillary First Molar Site. Compend Contin Educ Dent 2021;42:e1-e4.

Want to stay up to date?
youTooth.com is THE PLACE TO BE IN DENTISTRY – subscribe now and receive our monthly newsletter on top hot topics from the world of modern dentistry.